🔥 WhatsApp Group
Join now & get instant job alerts!
📢 Telegram Group
Don’t miss daily fresher job updates!
🚀 WhatsApp Channel
1-click join for verified job alerts!
Ipca Laboratories Limited is a leading fully integrated pharmaceutical company committed to manufacturing high-quality Active Pharmaceutical Ingredients (APIs) and formulations. With a strong presence across domestic and international markets, Ipca has established itself as a trusted name in the healthcare industry.
The company’s operations are supported by world-class manufacturing facilities approved by major regulatory bodies such as WHO, MHRA, and USFDA. Ipca focuses on innovation, stringent quality standards, and sustainable practices to deliver affordable and reliable healthcare solutions. With a legacy of excellence and continuous growth, Ipca Laboratories continues to create opportunities for professionals to build rewarding careers in various domains including Production, Quality Control, Quality Assurance, and Engineering.
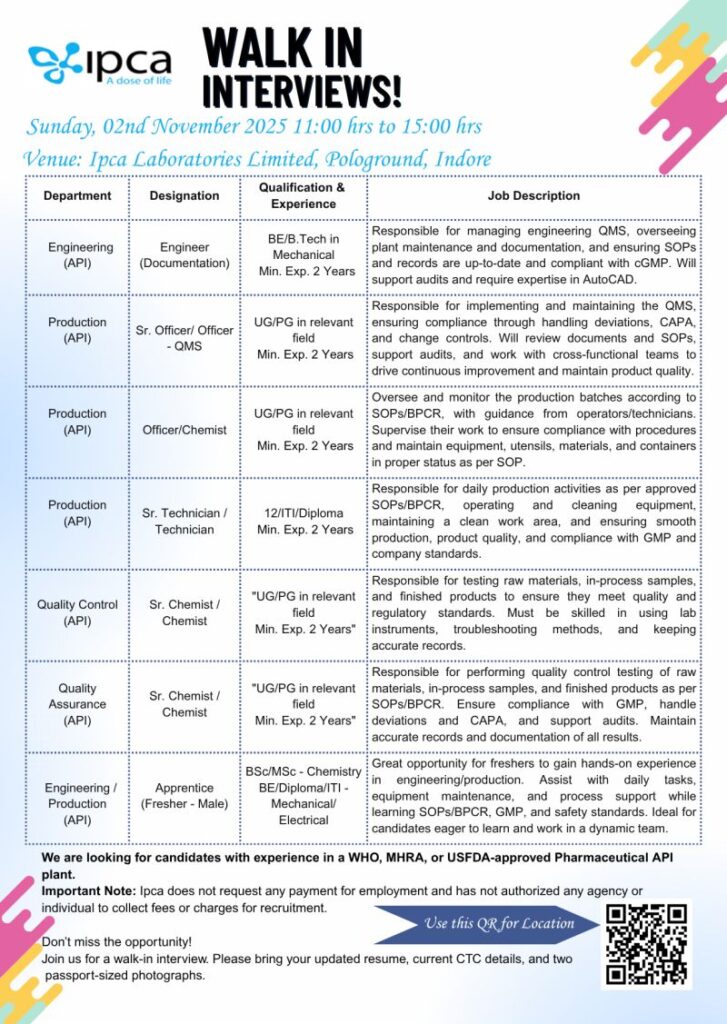
Company Name: Ipca Laboratories Limited
Date & Time: Sunday, 02nd November 2025 | 11:00 hrs to 15:00 hrs
Venue: Ipca Laboratories Limited, Pologround, Indore
Departments & Positions:
- Engineering (API): Engineer (Documentation)
- Production (API): Sr. Officer / Officer – QMS
- Production (API): Officer / Chemist
- Production (API): Sr. Technician / Technician
- Quality Control (API): Sr. Chemist / Chemist
- Quality Assurance (API): Sr. Chemist / Chemist
- Engineering / Production: Apprentice (Fresher – Male)
Qualification & Experience:
- BE/B.Tech in Mechanical Engineering
- UG/PG in relevant field with minimum 2 years of experience
- 12th / ITI / Diploma in relevant discipline
- BSc / MSc in Chemistry or BE / Diploma / ITI in Mechanical / Electrical for Apprentice
Job Responsibilities Include:
- Engineering QMS management, plant documentation & AutoCAD expertise
- QMS implementation, deviation handling & CAPA management
- Monitoring production batches as per SOPs/BPCR
- QA/QC of raw materials, in-process, and finished products
- Supporting audits, compliance with GMP & regulatory standards
- Hands-on learning for freshers in engineering/production
Who Can Apply:
Candidates with experience in WHO, MHRA, or USFDA-approved Pharmaceutical API plants are preferred.
Important Note:
Ipca Laboratories does not charge any fees for employment and has not authorized any individual or agency to collect such charges.
Documents to Carry:
Updated resume, current CTC details, and two passport-size photographs.