🔥 WhatsApp Group
Join now & get instant job alerts!
📢 Telegram Group
Don’t miss daily fresher job updates!
🚀 WhatsApp Channel
1-click join for verified job alerts!
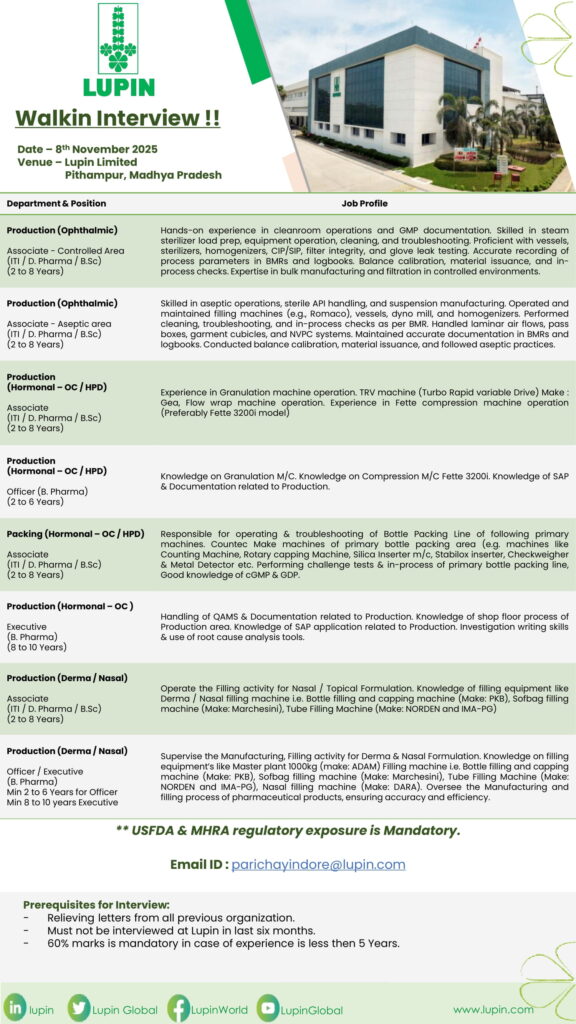
Company: Lupin Limited
Venue: Pithampur, Madhya Pradesh
Date: 8th November 2025
Latest Jobs
About the Company:
Lupin Limited is one of India’s leading multinational pharmaceutical companies with a strong global presence. The company focuses on the manufacturing and development of high-quality formulations, APIs, and specialty products across various therapeutic areas. Lupin is recognized for its commitment to innovation, regulatory excellence, and world-class manufacturing facilities compliant with USFDA and MHRA standards.
Departments & Positions:
Production (Ophthalmic / Hormonal / Derma / Nasal)
Positions: Associate / Officer / Executive
Qualification: ITI / D.Pharm / B.Pharm / B.Sc / M.Pharm
Experience: 2 to 10 Years
Job Description:
- Hands-on experience in sterile manufacturing and cleanroom operations.
- Knowledge of steam sterilizers, homogenizers, CIP/SIP systems, and filtration.
- Expertise in aseptic operations, granulation, compression, and filling (bottle, tube, nasal).
- Exposure to machines like Romaco, Gea, Fette 3200i, Marchesini, Norden, ADAM, and DARA.
- Experience in SAP, QAMS, and documentation as per cGMP & GDP standards.
Quality Control (Hormonal / OC / HPD / MDI / DPI / Nasal / Derma / Sterile)
Positions: Officer / Executive
Qualification: B.Pharm / M.Pharm / M.Sc / Any Graduate (Service Engineer)
Experience: 2 to 10 Years
Job Description:
- Proficient in HPLC, GC, LCMS, UV Spectrophotometer, and analytical instruments.
- Responsible for testing raw materials, intermediates, and finished products.
- Handling of OOS/OOT investigations, method validation, and calibration activities.
- Documentation & compliance with GMP/GLP standards and regulatory guidelines (USFDA, MHRA, EU).
- Experience in equipment calibration, vendor audits, and laboratory investigations.
Quality Assurance (API / Sterile)
Positions: Officer / Executive
Qualification: B.Pharm / M.Sc
Experience: 2 to 10 Years
Job Description:
- Review of BMRs/BPRs, CAPA, change control, and deviation management.
- Handling validation & qualification documentation.
- QA oversight in production and sampling activities.
- Preparation of SOPs, Master Plans, and audit readiness documentation.
- Involvement in regulatory submissions and compliance audits.
Validation / Microbiology (Sterile)
Positions: Officer / Executive
Qualification: B.Pharm / M.Sc (Microbiology)
Experience: 2 to 6 Years
Job Description:
- Conduct cleaning, equipment, and process validation.
- Experience in environmental monitoring, water testing, and microbiological analysis.
- Expertise in Autoclave, LAF, particle counter, and sterilization validation.
- Knowledge of cGMP, GDP, and regulatory compliance for sterile manufacturing.
Regulatory Exposure:
USFDA & MHRA regulatory exposure is mandatory.
Email for Application:
Prerequisites for Interview:
- Relieving letters from previous organization are mandatory.
- Candidates should not have attended any Lupin interview in the last 6 months.
- Minimum 60% marks required if experience is less than 5 years.