🔥 WhatsApp Group
Join now & get instant job alerts!
📢 Telegram Group
Don’t miss daily fresher job updates!
🚀 WhatsApp Channel
1-click join for verified job alerts!
About Company
Innoxel Lifesciences is a rapidly growing pharmaceutical company focused on delivering high-quality sterile and injectable products. The company is known for its strong compliance with global regulatory standards, including USFDA, and offers excellent career growth opportunities for pharma professionals.
Quick Details
- Company Name: Innoxel Lifesciences
- Job Type: Full-Time
- Qualification: B.Sc / M.Sc / B.Pharm / M.Pharm / ITI / Diploma / B.Com
- Experience: 1 to 10 Years (Sterile USFDA Experience Preferred)
- Location: India
- Selection Process: Shortlisting & Interview
Job Summary
Innoxel Lifesciences is inviting applications from experienced candidates for multiple departments including Quality Control, Sterile Manufacturing, QC Micro, Quality Assurance, Warehouse, Validation, Engineering, and Project Management. Candidates with sterile and USFDA exposure will be preferred.
Available Positions
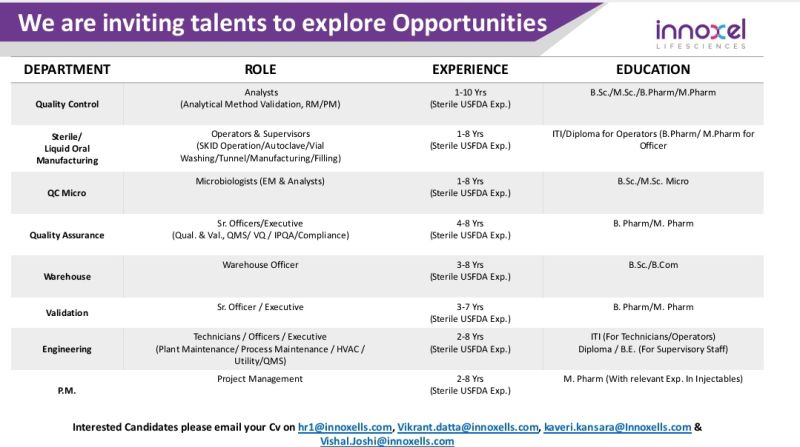
Quality Control
- Analyst (Analytical Method Validation, RM/PM)
- Experience: 1 to 10 Years
Sterile / Liquid Oral Manufacturing
- Operators & Supervisors
(SKID Operation / Autoclave / Vial Washing / Tunnel / Manufacturing / Filling) - Experience: 1 to 8 Years
QC Microbiology
- Microbiologists (EM & Analysts)
- Experience: 1 to 8 Years
Quality Assurance
- Sr. Officer / Executive
(Qualification & Validation, QMS, IPQA, Compliance) - Experience: 4 to 8 Years
Warehouse
- Warehouse Officer
- Experience: 3 to 8 Years
Validation
- Sr. Officer / Executive
- Experience: 3 to 7 Years
Engineering
- Technicians / Officers / Executives
(Plant Maintenance, Process Maintenance, HVAC, Utility, QMS) - Experience: 2 to 8 Years
Project Management (P.M.)
- Role: Project Management
- Experience: 2 to 8 Years
Job Criteria
- Candidates must have relevant pharmaceutical experience
- Preference will be given to candidates with sterile USFDA exposure
- Good technical knowledge and documentation skills required
- Immediate joiners are preferred
How to Apply
Interested candidates can send their updated CV to:
Important Note
- Mention your department and position in the subject line
- Ensure your resume is updated with relevant experience
FAQ
Q1. Who can apply?
Candidates with 1 to 10 years of relevant pharma experience can apply.
Q2. Is USFDA experience mandatory?
Preferred, especially for sterile-related roles.
Q3. Are freshers eligible?
No, experienced candidates are required.
Q4. Which departments are hiring?
Quality Control, Manufacturing, QC Micro, QA, Warehouse, Validation, Engineering, and Project Management.