🔥 WhatsApp Group
Join now & get instant job alerts!
📢 Telegram Group
Don’t miss daily fresher job updates!
🚀 WhatsApp Channel
1-click join for verified job alerts!
About Company
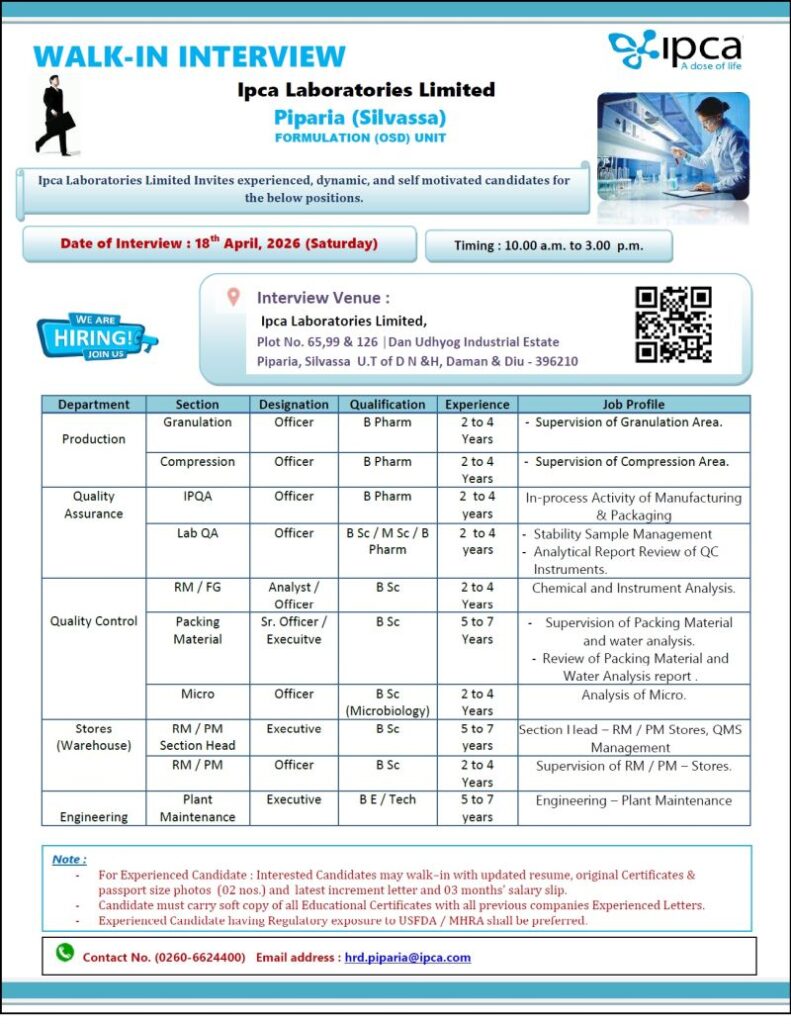
Ipca Laboratories Limited is a well-established pharmaceutical company known for its strong presence in API and formulation manufacturing. The company is committed to quality, innovation, and global regulatory standards, offering excellent career opportunities for skilled professionals in the pharma sector.
Interview Date & Time
Date: 18 April 2026 (Saturday)
Time: 10:00 AM to 03:00 PM
Job Location
Piparia (Silvassa) – Formulation (OSD) Unit
Interview Venue
Ipca Laboratories Limited
Plot No. 65, 98 & 126,
Dan Udyog Industrial Estate,
Piparia, Silvassa (UT of DNH & DD) – 396210
Available Departments & Job Details
Production
Granulation / Compression
- Qualification: B.Pharm
- Designation: Officer
- Experience: 2 to 4 Years
- Job Role: Supervision of granulation & compression areas
Quality Assurance
IPQA
- Qualification: B.Pharm
- Experience: 2 to 4 Years
- Role: In-process manufacturing & packaging activities
Lab QA
- Qualification: B.Sc / M.Sc / B.Pharm
- Experience: 2 to 4 Years
- Role: Stability sample management, analytical report review
Quality Control
RM / FG (Analyst / Officer)
- Qualification: B.Sc
- Experience: 2 to 4 Years
- Role: Chemical & instrument analysis
Packing Material (Sr. Officer / Executive)
- Qualification: B.Sc
- Experience: 5 to 7 Years
- Role: Packing material & water analysis, report review
Microbiology
- Qualification: B.Sc (Microbiology)
- Experience: 2 to 4 Years
- Role: Microbial analysis
Stores (Warehouse)
RM / PM Section Head (Executive)
- Qualification: B.Sc
- Experience: 5 to 7 Years
- Role: Stores handling, QMS management
RM / PM (Officer)
- Qualification: B.Sc
- Experience: 2 to 4 Years
- Role: Supervision of RM/PM stores
Engineering
Plant Maintenance
- Qualification: B.E / B.Tech
- Experience: 5 to 7 Years
- Role: Plant maintenance operations
Eligibility Criteria
- Candidates must have relevant qualifications and experience
- Exposure to regulatory audits like USFDA / MHRA will be preferred
Documents Required
- Updated Resume
- Original Educational Certificates
- Passport Size Photos (2 copies)
- Last Increment Letter
- Last 3 Months Salary Slips
- Previous Experience Certificates
How to Apply
Interested candidates can directly walk in for the interview with required documents.
Contact Details
Phone: 0260-6624400
Email: hrd.piparia@ipca.com
Note
- Only experienced candidates are eligible
- Candidates must carry all original documents and supporting proofs