🔥 WhatsApp Group
Join now & get instant job alerts!
📢 Telegram Group
Don’t miss daily fresher job updates!
🚀 WhatsApp Channel
1-click join for verified job alerts!
About Company:
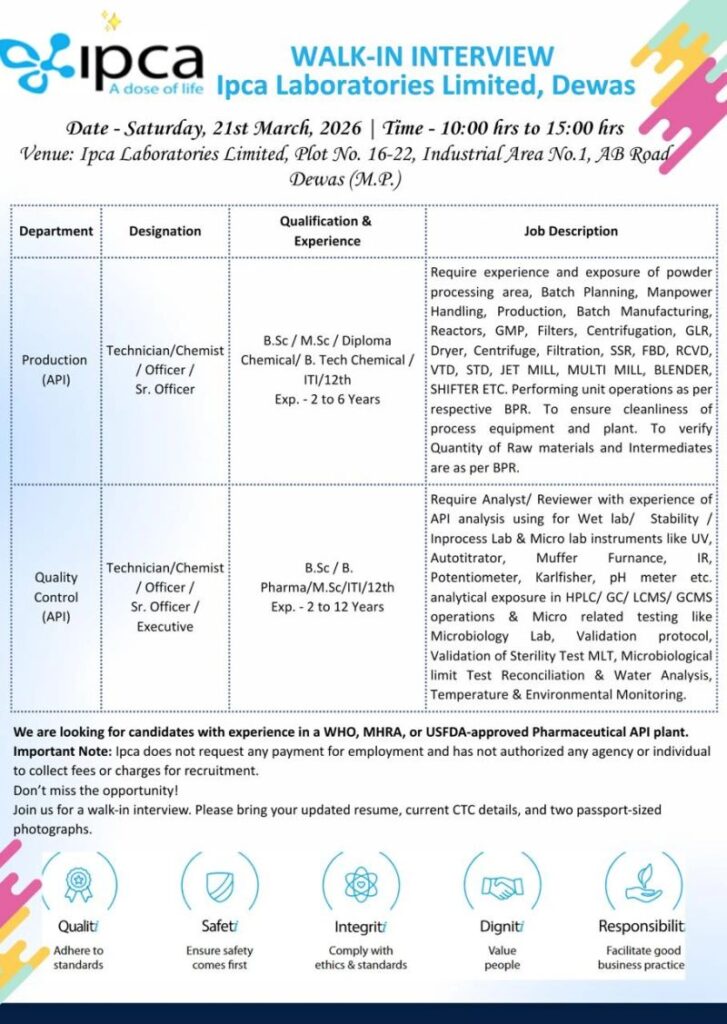
Ipca Laboratories Limited is a leading global pharmaceutical company with a strong presence in over 120 countries. The company is well known for manufacturing high-quality Active Pharmaceutical Ingredients (API) and finished dosage formulations. Ipca operates several world-class manufacturing facilities approved by international regulatory authorities such as WHO, MHRA, and USFDA. With a strong focus on innovation, quality, and compliance, Ipca continues to deliver affordable and effective medicines worldwide.
Interview Date & Time:
21 March 2026 (Saturday)
10:00 AM to 03:00 PM
Interview Venue:
Ipca Laboratories Limited
Plot No. 16–22, Industrial Area No.1,
AB Road, Dewas, Madhya Pradesh
Department:
Production (API)
Designation:
Technician / Chemist / Officer / Sr. Officer
Qualification:
B.Sc / M.Sc / Diploma Chemical / B.Tech Chemical / ITI / 12th
Experience:
2 to 6 Years
Job Responsibilities:
Candidates should have experience in powder processing area, batch planning, manpower handling, production, and batch manufacturing. Experience in handling reactors, filters, centrifugation, GLR, dryer, centrifuge, filtration, SSR, FBD, RCVD, VTD, STD, jet mill, multi mill, blender, and shifter is preferred. Candidates should be able to perform unit operations as per BPR, ensure cleanliness of process equipment and plant, and verify the quantity of raw materials and intermediates as per BPR.
Department:
Quality Control (API)
Designation:
Technician / Chemist / Officer / Sr. Officer / Executive
Qualification:
B.Sc / B.Pharm / M.Sc / ITI / 12th
Experience:
2 to 12 Years
Job Responsibilities:
Candidates should have experience in API analysis for Wet Lab, Stability, In-process Lab, and Micro Lab. Knowledge of instruments such as UV, Autotitrator, Muffle Furnace, IR, Potentiometer, Karl Fischer, and pH meter is required. Experience in analytical instruments such as HPLC, GC, LCMS, and GCMS is preferred. Candidates should also have exposure to microbiology testing, validation protocols, sterility testing, microbial limit testing, water analysis, and environmental monitoring.
Note:
Candidates with experience in WHO, MHRA, or USFDA approved pharmaceutical API plants will be preferred.
Documents Required:
- Updated Resume
- Current CTC Details
- Two Passport Size Photographs
Candidates are advised to attend the walk-in interview with the required documents. Ipca Laboratories Limited does not charge any fees for recruitment and has not authorized any agency to collect charges for employment.